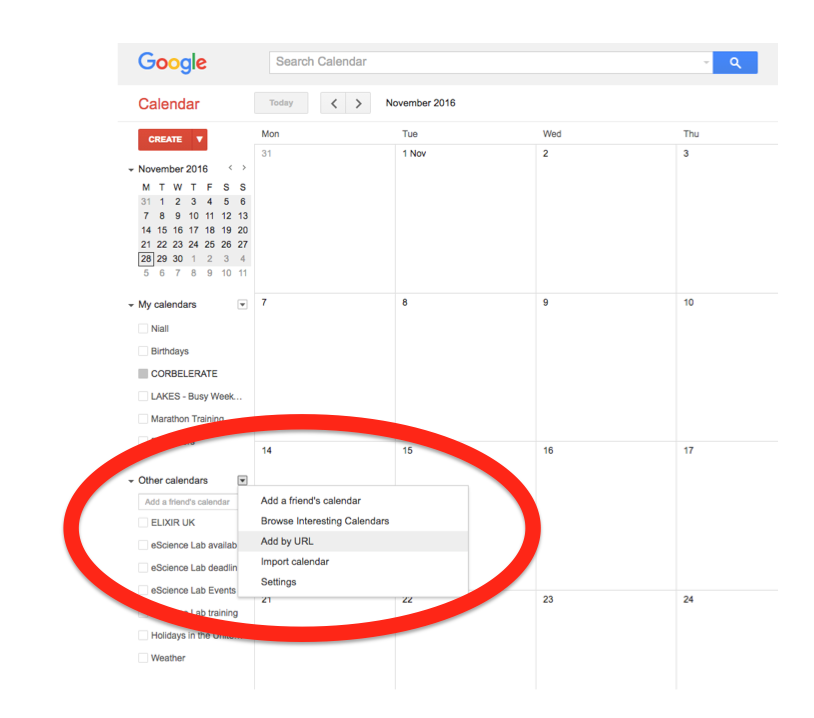
Events
Include expired: true
and Keywords: Process Validation or CFR 820 or Guidance
and City: Berlin or Bogota or Copenhagen or La Jolla or Liverpool or Philadelphia or Toronto
and Target audience: Medical Device or Process Development or Regulatory Affairs